Clinical Docs Library
Guides, templates, and regulatory frameworks for clinical trial documentation. Written by medical writers with 15+ years in pharma.
A Guide to Regulatory Affairs
Overview of regulatory affairs in clinical trials — key agencies, documentation standards, and submission requirements for pharma teams.
Audit Trails in Clinical Trial Documentation
How to implement compliant audit trails in clinical documentation — FDA 21 CFR Part 11, eTMF requirements, and best practices.
Document Traceability in Clinical Trials
Document traceability frameworks for clinical trials — linking evidence to claims, cross-document consistency, and regulatory audit readiness.
Common Documentation Findings in Regulatory Inspections
Most common clinical documentation findings during FDA and EMA inspections — what inspectors look for and how to avoid deficiencies.
EU CTR and Documentation Workflows
EU Clinical Trials Regulation documentation workflows — submission requirements, CTIS system, and practical implementation guidance.
FDA 21 CFR Part 11 Implications for Clinical Documentation
Practical implications of FDA 21 CFR Part 11 for clinical trial documentation — electronic records, signatures, and audit trail requirements.
Automate the documents you're reading about
Skaldi generates audit-ready clinical trial documents in minutes, not weeks.
eTMF Governance Models
eTMF governance frameworks for clinical trials — roles, responsibilities, access controls, and quality oversight models.
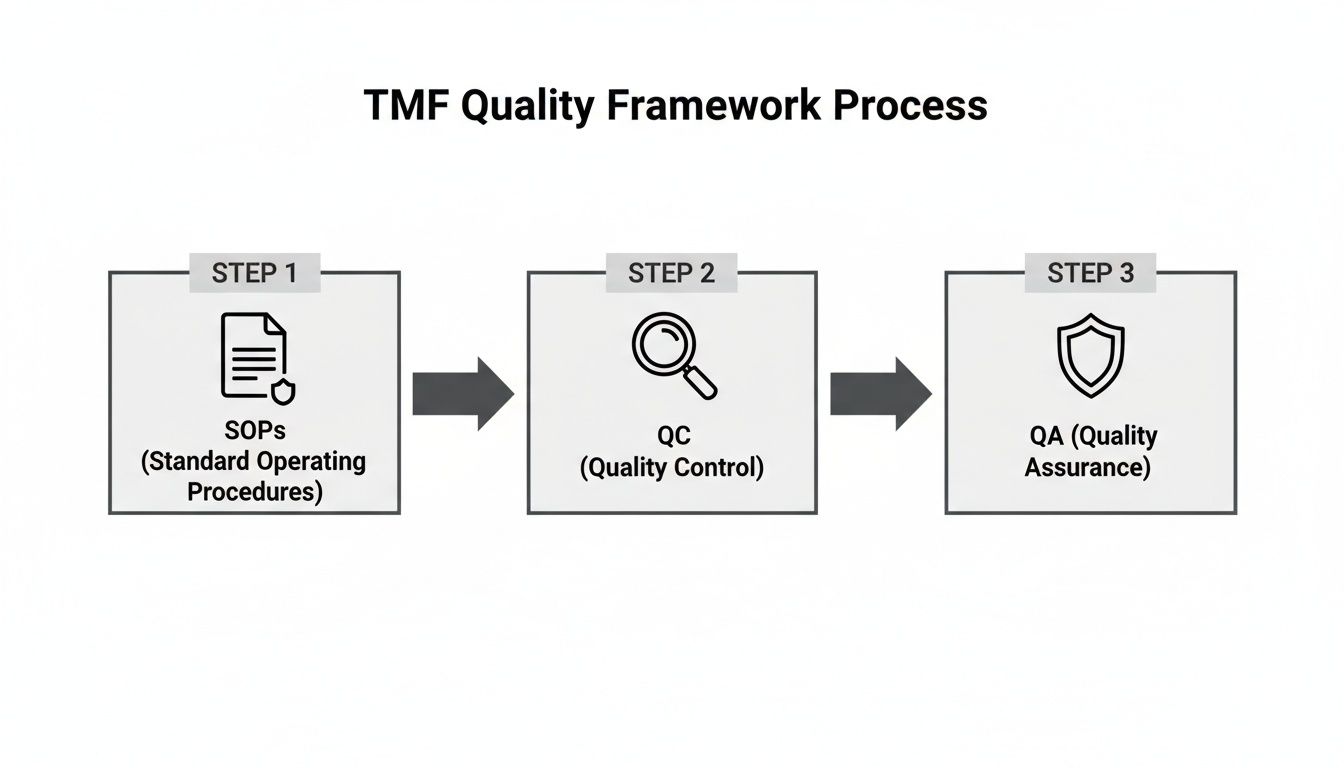
TMF Quality Control
TMF quality control strategies — inspection readiness metrics, QC checklists, and common TMF deficiencies to avoid.

Trial Master File Completeness
How to achieve and maintain TMF completeness — DIA TMF Reference Model alignment, completeness metrics, and inspection readiness.
Clinical Study Report Authoring and Version Management
CSR authoring workflow and version management — ICH E3 structure, co-authoring best practices, and regulatory submission formatting.
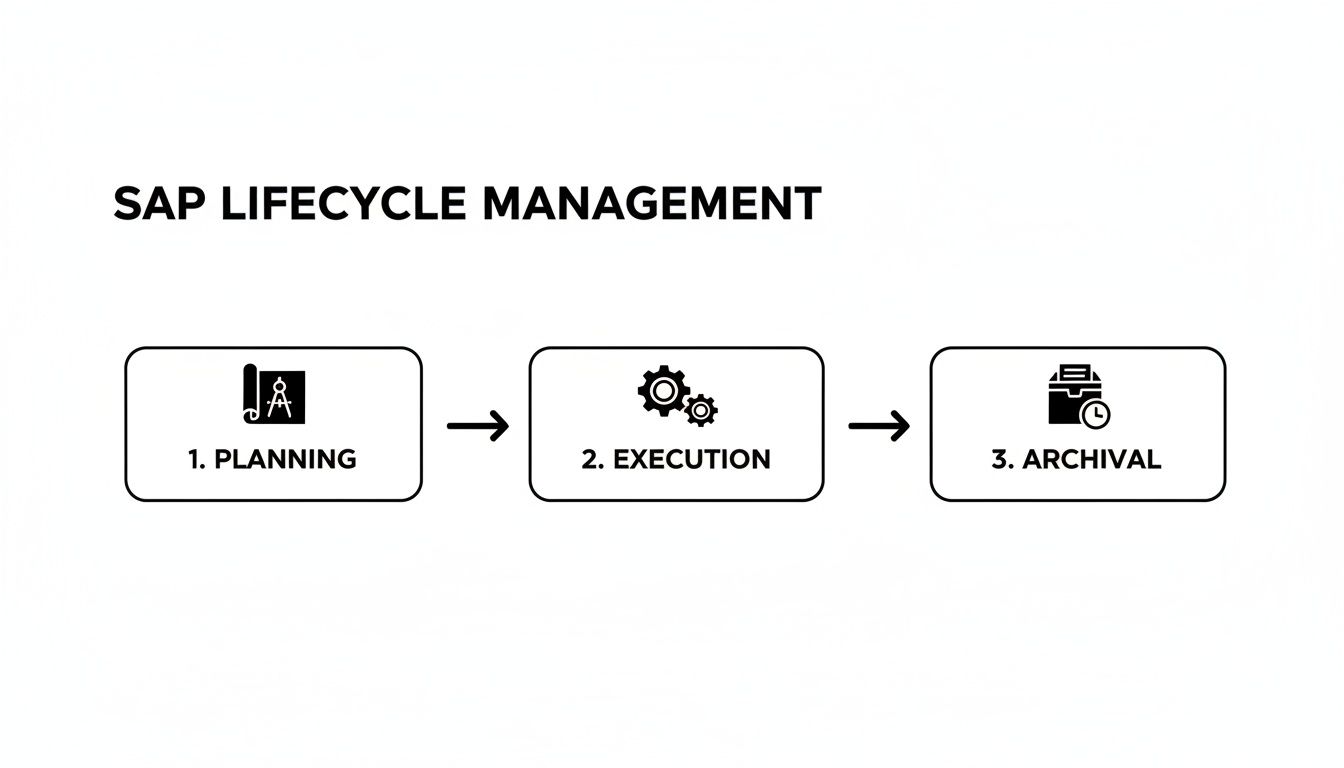
SAP Lifecycle Management
Statistical Analysis Plan lifecycle management — from protocol-aligned pre-specification through amendments and final SAP sign-off.
Mastering IB Updates
How to manage Investigator's Brochure updates — trigger criteria, update cycles, version control, and regulatory notification requirements.
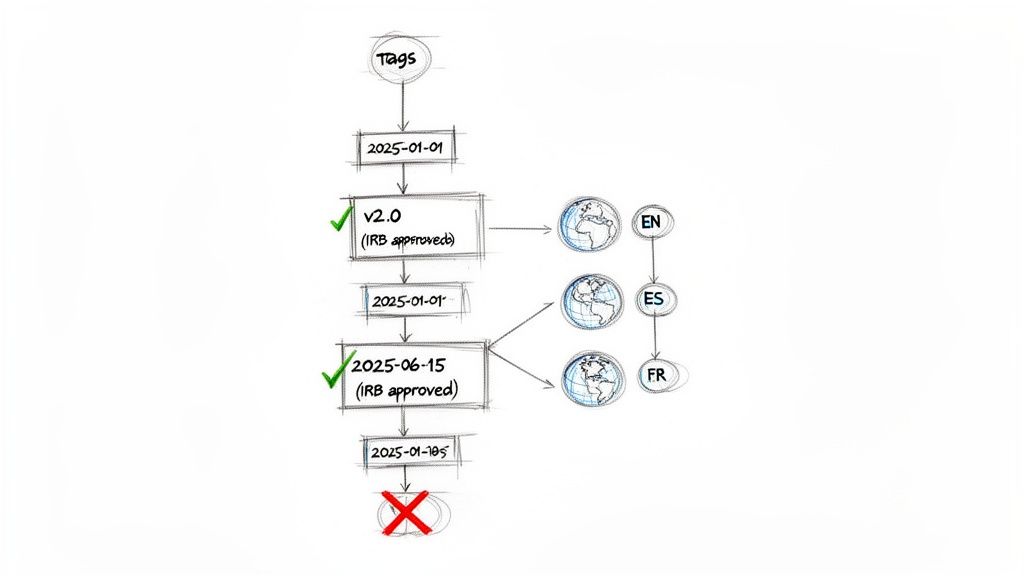
Managing Informed Consent Updates
Managing ICF updates during an ongoing trial — triggers, re-consent procedures, IRB/IEC requirements, and documentation standards.

RACI Model for Clinical Documentation
RACI model for clinical documentation — how to assign roles and responsibilities across medical writing, clinical, and regulatory teams.
Document Review and Approval Workflows
Clinical document review and approval workflows — routing, review cycles, comment resolution, and electronic approval best practices.
Version Control Best Practices
Version control best practices for clinical documents — naming conventions, change tracking, controlled copies, and audit trail requirements.
Change Control in Clinical Documentation
Change control in clinical documentation — when changes require formal control, impact assessment, and documentation update procedures.
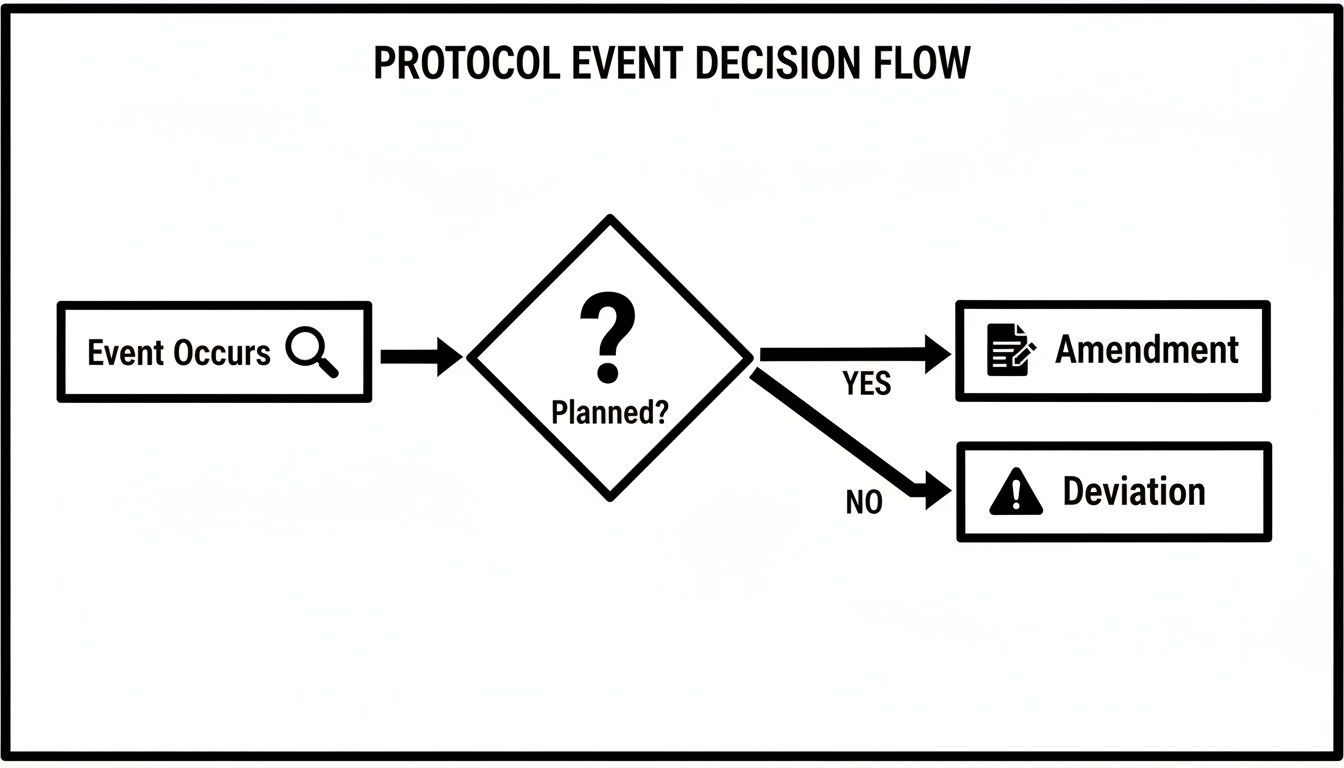
Protocol Deviations vs Amendments
Protocol deviations vs. amendments — definitions, classification criteria, reporting requirements, and documentation implications.
Managing Protocol Amendments
How to manage protocol amendments — substantial vs. non-substantial changes, regulatory submission, site notification, and TMF updates.
Clinical Trial Document Lifecycle Management
End-to-end clinical trial document lifecycle — from study design through archiving, with key milestones and handoff points.
Medical Writing for Clinical Trials
Medical writing fundamentals for clinical trials — document types, regulatory standards, quality principles, and career guidance for writers.
Pharmaceutical QMS
Pharmaceutical quality management systems for clinical trials — GCP compliance, document control, CAPA, and QMS implementation guidance.
A Practical Guide to the Trial Master File
Practical TMF guide — structure, essential documents, filing principles, and how to maintain inspection-ready trial master files.

Regulatory Documents in Clinical Trials
Overview of regulatory documents required in clinical trials — IND, CTA, IB, Protocol, CSR, and submission package requirements.

FDA 21 CFR Part 11 Compliance Guide
Step-by-step FDA 21 CFR Part 11 compliance guide — system validation, electronic signatures, audit trails, and common compliance gaps.
ICH Guidelines for Clinical Trials
ICH guidelines for clinical trials — E6(R3) GCP, E3 CSR, E8 study design, E9 statistics, and practical application for study teams.
Informed Consent Form Template
Informed consent form template with all required ICH E6 and FDA elements — structured sections, reading level guidance, and IRB tips.

SAP Template for Clinical Trials
Statistical Analysis Plan template for clinical trials — pre-specified analyses, estimands, missing data, and ICH E9(R1) alignment.
CSR Template
Clinical Study Report template aligned with ICH E3 — all required sections, appendix structure, and formatting guidance for submission.
Protocol Development for Clinical Trials
Clinical trial protocol development guide — study design, endpoints, eligibility criteria, procedures, and ICH E6(R3) alignment.
Clinical Trial Protocol Template
Clinical trial protocol template with all ICH E6(R2) required sections — study design, objectives, eligibility, procedures, and statistics.
DIA TMF Reference Model
DIA TMF Reference Model explained — zones, sections, artifacts, and how to map your eTMF structure to the reference model.

Electronic Trial Master File Software
How to evaluate and select electronic TMF software — key features, vendor checklist, validation requirements, and implementation tips.
Regulatory Document Management Systems
Regulatory document management systems for pharma — key capabilities, compliance requirements, and vendor evaluation criteria.
Want to see these documents generated automatically?